US In Vitro Diagnostics Market to Reach USD 57.18 Billion by 2031
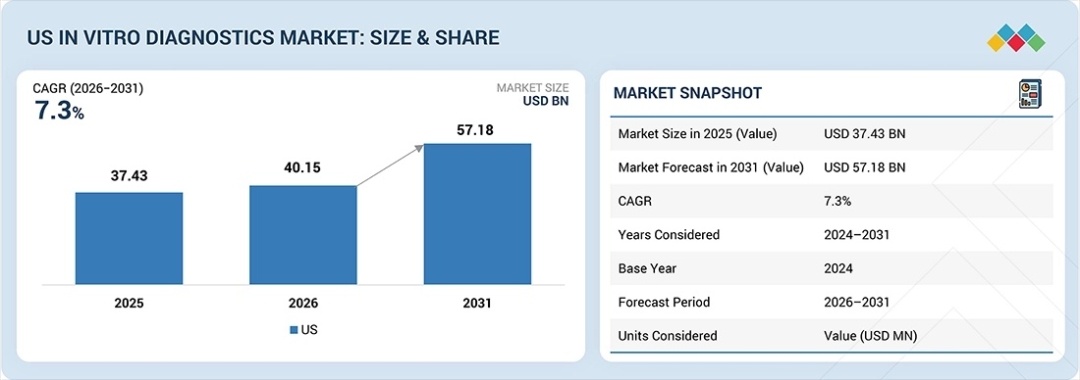
The US In Vitro Diagnostics (IVD) market continues to demonstrate steady expansion driven by increasing healthcare needs and rapid advancements in diagnostic technologies. Valued at USD 37.43 billion in 2025, the market reached USD 40.15 billion in 2026 and is projected to grow at a CAGR of 7.3% from 2025 to 2031, reaching an estimated USD 57.18 billion by 2031. The rising prevalence of chronic and infectious diseases across the country is one of the primary forces fueling demand for advanced diagnostic solutions. Healthcare providers increasingly rely on accurate and early diagnostic testing to guide treatment decisions, improve patient outcomes, and reduce healthcare costs.
Download PDF Brochure:https://www.marketsandmarkets.com/pdfdownloadNew.asp?id=66290979
The expansion of the IVD market is further supported by the growing adoption of molecular diagnostic technologies, point-of-care testing solutions, and automated laboratory systems. Continuous innovations in diagnostic instruments, reagents, and assays are improving testing efficiency and diagnostic accuracy across healthcare facilities. Additionally, the aging population in the US is significantly increasing testing demand due to the higher incidence of chronic diseases among older adults. Expanding laboratory infrastructure, improvements in healthcare access, and regulatory support for diagnostic innovation are also playing key roles in accelerating market growth across hospitals, diagnostic laboratories, and research facilities.
Rising Geriatric Population and Chronic Disease Burden Driving Market Demand
The rapidly growing geriatric population in the United States is significantly contributing to the increased demand for in vitro diagnostic testing. Older adults are more susceptible to chronic diseases such as diabetes, cardiovascular diseases, cancer, and respiratory disorders, all of which require continuous monitoring and early detection. As the population aged 65 and above continues to expand, healthcare systems are experiencing a growing need for routine screening, preventive diagnostics, and disease monitoring solutions. This demographic shift is driving higher testing volumes across hospitals, clinics, and clinical laboratories nationwide.
Chronic disease management relies heavily on accurate diagnostic testing to detect disease progression and guide treatment strategies. For example, diabetes patients require frequent monitoring of blood glucose levels and related biomarkers, while cardiovascular disease patients depend on cardiac biomarker testing and lipid profiling. Similarly, oncology diagnostics increasingly utilize molecular testing and biomarker-based assays to support precision medicine and personalized therapies. As healthcare providers emphasize early disease detection and preventive care, the demand for reliable IVD solutions continues to increase.
The availability of advanced diagnostic technologies has also strengthened the role of laboratories and healthcare institutions in disease surveillance and management. Automated analyzers, high-throughput testing systems, and integrated data platforms allow laboratories to process large volumes of samples efficiently while maintaining accuracy and quality. Consequently, the expanding elderly population and the increasing burden of chronic diseases are expected to remain key drivers supporting long-term growth of the US IVD market.
Technological Innovation and Biomarker Development Unlock New Opportunities
Continuous advancements in diagnostic technologies are creating new opportunities for growth in the US IVD market. One of the most significant developments is the introduction of disease-specific biomarkers and companion diagnostics, which enable earlier detection of diseases and facilitate personalized treatment decisions. These innovations are particularly impactful in areas such as oncology, cardiology, infectious diseases, and neurodegenerative disorders.
Biomarker-driven diagnostics are transforming clinical decision-making by providing physicians with precise insights into disease progression, patient response to therapies, and risk stratification. For instance, oncology diagnostics increasingly rely on biomarker testing to identify specific genetic mutations that can guide targeted therapy selection. Similarly, cardiovascular diagnostics utilize biomarker-based tests to detect heart damage and predict patient risk, enabling faster clinical interventions.
In addition to biomarker advancements, improvements in molecular diagnostics, immunoassays, and high-throughput laboratory systems are further enhancing testing accuracy and efficiency. The integration of automation and artificial intelligence in diagnostic workflows is also improving laboratory productivity and reducing turnaround times. Point-of-care testing technologies are expanding diagnostic access by enabling rapid testing outside traditional laboratory environments, including emergency departments, clinics, and remote healthcare settings.
The introduction of innovative testing platforms is also driving the demand for reagents and diagnostic kits, which remain essential components of routine and specialized diagnostic workflows. Reagents and kits represented the largest product segment in 2025 due to their continuous consumption across clinical laboratories performing molecular diagnostics, immunoassays, and clinical chemistry testing. Increasing screening programs and rising test volumes are expected to sustain strong demand for these consumables throughout the forecast period.
Request Sample Pages-https://www.marketsandmarkets.com/requestsampleNew.asp?id=66290979
Market Challenges and Segment Trends Shaping Future Growth
Despite strong growth prospects, the US IVD market faces several operational and economic challenges. One of the most significant barriers is the high cost associated with advanced diagnostic equipment, including molecular analyzers, automated laboratory platforms, and high-throughput testing systems. These technologies often require substantial capital investment, making adoption difficult for small and mid-sized laboratories. Additional costs related to maintenance, consumables, quality compliance, and skilled personnel further increase operational expenses.
Operational complexities also present ongoing challenges for market participants. Diagnostic companies and laboratories must navigate complex regulatory approval processes, ensure compliance with quality standards, and manage documentation requirements associated with laboratory operations. Variability in reimbursement policies across healthcare systems can also affect the adoption of new diagnostic tests. Furthermore, workforce shortages in clinical laboratories and difficulties in integrating advanced diagnostic platforms into existing workflows may slow the implementation of innovative technologies.
From a segment perspective, immunoassays continue to dominate the technology landscape due to their broad applicability across infectious disease testing, endocrinology, oncology, and chronic disease monitoring. Their high analytical sensitivity, rapid turnaround times, and compatibility with automated platforms make them indispensable in modern diagnostic laboratories. Blood, serum, and plasma remain the most widely used specimen types due to their compatibility with multiple diagnostic modalities.
Centralized laboratory testing continues to represent the dominant site of testing within the US IVD market. Well-established laboratory infrastructure, advanced analytical capabilities, and the need for complex diagnostic testing support the strong presence of centralized laboratories. Meanwhile, infectious disease testing remains the largest application area, driven by ongoing screening programs for conditions such as HIV, hepatitis, tuberculosis, and respiratory infections.
Hospitals and clinics represent the largest end-user segment due to high patient volumes and the integration of advanced diagnostic platforms within routine clinical workflows. These facilities rely heavily on molecular and immunoassay analyzers to support emergency care, inpatient diagnostics, and chronic disease management. Increasing automation and a focus on improving diagnostic efficiency are expected to further strengthen their position in the market.
Key companies operating in the US IVD market include Danaher (US), F. Hoffmann-La Roche Ltd. (Switzerland), Abbott (US), Siemens Healthineers AG (Germany), and Thermo Fisher Scientific, Inc. (US), all of which continue to invest heavily in research, technological innovation, and strategic collaborations to strengthen their market presence.
Related Markets
Media Contact
Company Name: MarketsandMarkets™ Research Private Ltd.
Contact Person: Mr. Rohan Salgarkar
Email: Send Email
Phone: 18886006441
Address:1615 South Congress Ave. Suite 103, Delray Beach, FL 33445
City: Florida
State: Florida
Country: United States
Website: https://www.marketsandmarkets.com/Market-Reports/u-s-in-vitro-diagnostics-market-66290979.html
Press Release Distributed by ABNewswire.com
To view the original version on ABNewswire visit: US In Vitro Diagnostics Market to Reach USD 57.18 Billion by 2031
Information contained on this page is provided by an independent third-party content provider. XPRMedia and this Site make no warranties or representations in connection therewith. If you are affiliated with this page and would like it removed please contact [email protected]