An Imaging Revolution Begins: Forbes Elite Xiaoyu Zhao is Mapping the 4D Future of Medicine
In the multibillion-dollar race to develop next-generation medicines, the industry has hit a "visibility ceiling." While we can sequence a genome in hours, we remain remarkably blind to the spatial metabolism that determines whether a drug actually works. Xiaoyu Zhao, founder of SuperVision Medicine, is finally turning the lights on.
In the high-stakes world of drug development, there is a $200 billion problem that few talk about: the "blind spot." Despite advances in AI and genomics, roughly 90% of clinical trials still end in failure. The reason is often a fundamental disconnect between the lab and the living body. Today, biopharma is attempting to build the next generation of therapies using 'decoy' data—images of tissue that have been processed to the point of chemical erasure, losing the critical signals that determine a drug's success.

“Traditional pathology is, in many ways, a process of destruction,” says Xiaoyu (Katherine) Zhao, the 25-year-old Chair and CEO of SuperVision Medicine. “To look at a tumor under a microscope, scientists traditionally fix it in wax and soak it in solvents. By the time you see the image, the most vital clues—volatile metabolites and delicate lipids—have been washed away.”
Zhao, recently spotlighted in the 2025 Forbes China 100 Returnee Elites Selection, calls these traditional samples "chemical ghosts." Her mission is to replace these ghosts with a "living map" through a framework she calls “Ramanomics”—a label-free, 4D molecular lens designed to capture the chemical heartbeat of life in its native state, without the legacy of destruction.

The Super-Multiplex Revolution: Seeing Biology in Its Native State
In modern R&D, molecular imaging has long been defined by a zero-sum game: researchers can either maximize molecular detail or preserve a tissue’s native state, but rarely both. Mass Spectrometry Imaging (MSI), the industry’s traditional "bridge," remains a destructive and laborious process, typically constrained to a "blurry" 10–50μm resolution. In the high-stakes world of cellular signaling, a 10-micron gap is more than a technical footnote—it is a canyon that swallows the very sub-cellular boundaries where diseases are won or lost.
SuperVision Medicine shatters this compromise by leveraging the intrinsic physics of Raman scattering to bypass the industry’s greatest bottleneck: sample preparation. While sub-micron resolution (<1μm) is a natural property of raman light, the true breakthrough lies in eliminating the "legacy of destruction." Traditional high-dimensional imaging requires "fixing" tissue in wax or harsh solvents—a process of chemical erasure that distorts the very metabolism researchers need to see.
By removing the need for fixation, SuperVision delivers a 50x increase in spatial resolution compared to standard MSI, applied directly to fresh, living specimens. This enables a "Super-Multiplexed" view where the Extracellular Matrix (ECM), carbohydrate and lipid metabolites, sterols, and drug compounds are visualized simultaneously within a single, unified 4D coordinate system.
This is not merely a sharper image; it is a fundamental shift in the operating model of drug discovery. Conventional workflows are inherently fragmented—requiring one tissue slice for drug distribution, another for the ECM, and a third for metabolites—consuming precious clinical samples while breaking the spatial links that make these signals interpretable. Importantly, the ECM encodes critical signaling information during tissue or organoid development, yet current methods cannot achieve in-situ labeling. Conventional approaches require ECM removal and molecular isolation, which inevitably leads to loss of spatial context. Zhao’s solution uniquely preserves both spatial and temporal information of extracellular signaling, including exosomal communication within the ECM—an unmet challenge that has never before been addressed in the field.
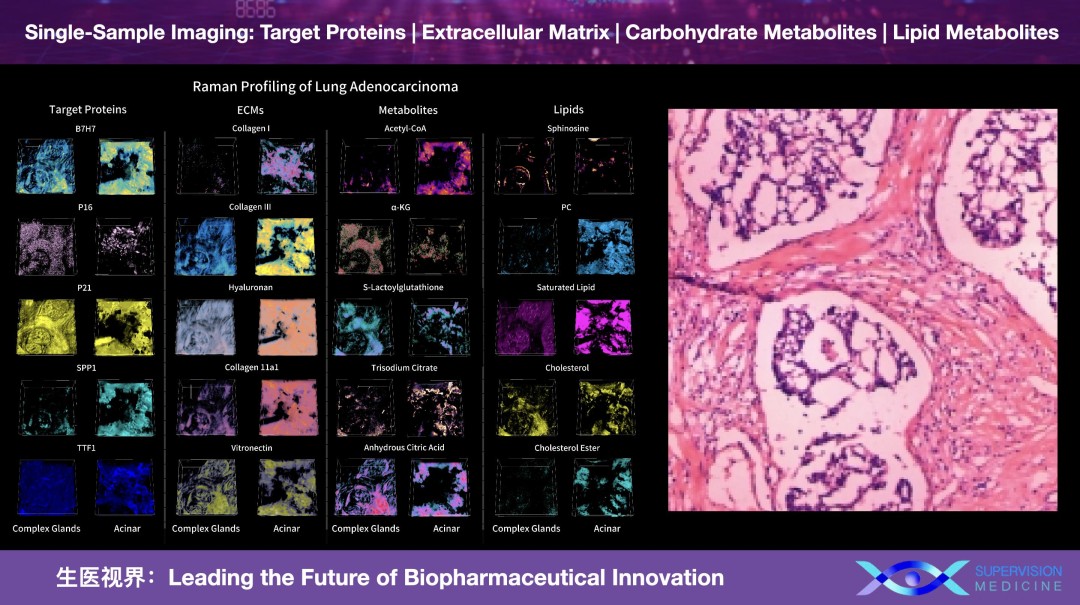
SuperVision Medicine "changes the math" by reading intrinsic vibrational fingerprints to capture these diverse marker classes from a single fresh sample. This preserves both the spatial context and the immense value of the specimen. For surgeons in the operating room and pharma R&D teams in the lab, this "sample-first" approach turns a patchwork of partial readouts into a single actionable map, supporting faster, higher-confidence decisions without sacrificing the biological integrity of the tissue.
Transforming High-End Physics into Actionable R&D Intelligence
Recognizing that high-end Raman systems are expensive "laser physics labs" that most pharma teams aren't equipped to run, Zhao has scaled access via a disruptive Imaging-as-a-Service model. SuperVision acts as a "Navigation Layer" for R&D, allowing pharma giants to use fast, label-free Raman maps to identify "hotspots" before committing to expensive, destructive downstream assays.
Furthermore, unlike "black box" AI tools that rely on pattern recognition without underlying chemical proof, Ramanomics is chemically deterministic. It produces reproducible outputs based on immutable physical signatures—a critical requirement for clinical standardization and regulatory approval.
A Founder for the Deep-Tech Era
Born in 2000, Zhao represents a new generation of Gen-Z founders who prioritize efficiency over legacy. Her trajectory was shaped by a rare triad of mentors at the forefront of their fields: Dame Molly Stevens, a pioneer in biomedicine; Prof. Tom Vercauteren, a leader in clinical image computing; and Prof. Wei Min, whose work helped define stimulated Raman microscopy.
This rare intersection of bioengineering, clinical translation, and frontier optics instilled in Zhao a pragmatic business philosophy: the product is not the microscope; it is the multi-million dollar decision the microscope enables. Her work was recently validated in a 2025 Nature Photonics review, placing SuperVision Medicine alongside the world’s most established international Raman microscopy providers.

The Molecular Lens: Designing a Future Where Chemistry is Visible
From helping a pharmaceutical giant validate a metabolic inhibitor to assisting a surgeon in real-time tumor delineation, Zhao’s goal remains constant: If you can see the chemistry, you can change the course of disease. As the industry moves toward complex organoid models and personalized therapies, the "molecular lens" of SuperVision Medicine is shifting from a luxury to a necessity. In the race for the next medical breakthrough, the winners will be those who can finally see the finish line in 4D.
Information contained on this page is provided by an independent third-party content provider. XPRMedia and this Site make no warranties or representations in connection therewith. If you are affiliated with this page and would like it removed please contact [email protected]

